Playing hip-hop or Doris Day, older exercise instructors can speak their
peers' language
[June 10, 2026]
By DONNA BLANKINSHIP
SEATTLE (AP) — Bengie Santos was a dancer and choreographer for 30 years
before reaching an age when most artists hang up their dancing shoes and
take a seat in the audience. But she wasn’t ready to sit back and enjoy
the show. So Santos began her third act as an exercise instructor.
Now 72 and in her 14th year teaching at the YMCA of Greater Seattle,
Santos has an almost cult-like following of older adults — many in their
80s and 90s — who come to stretch, dance and lift weights multiple times
a week.
From Doris Day singalongs to hip-hop routines and country line dances,
the classes get people kicking, popping and spinning.
Some of Santos' students say younger instructors — however great — don’t
quite “get them” in the same way.
And Y organizers and other experts say many older instructors can be
role models, speaking their peers' language in a way that makes exercise
more fun.
Exercise can build social connections
Sharon Ruff, 81, was attracted to Santos’ classes by the eclectic mix of
music, and loves the instructor’s energy and enthusiasm. Before she
retired from her job as a teacher, she had never exercised regularly. “I
didn’t really like it, because it wasn’t fun,” she says.
Her friend Ann Kashiwa, 91, often stays for two hour-long classes in a
row. She started taking Santos’ classes during the pandemic, and has
stuck with them through treatment for pancreatic cancer.

“She is not only a fantastic teacher, but she gets people together,”
says Kashiwa, whose Y friendships have helped her through tough times.
Santos says inspiration flows both ways. Her students inspire her to
keep going, she says, and “I’m hoping I inspire them to keep going.”
What older instructors can offer
Exercise and being with people are crucial for healthy aging, so finding
incentives to do that is important.
“Everybody has to figure out what their barriers are” and come up with
strategies to make physical activity rewarding, says Daniel E.
Lieberman, an evolutionary biologist at Harvard and author of several
books, including “Exercised: Why Something We Never Evolved to Do is
Healthy and Rewarding.”
Finding someone to get you moving, whether an instructor or a friend,
can help overcome that natural resistance.
“People who stay active as they get older are important role models for
everyone,” Lieberman says.
Harry King, an 83-year-old personal trainer in Greenville, South
Carolina, agrees that motivation to take that first step is tough for
many people, and becomes even harder as their mobility weakens.
King had always been active – kayaking, hiking and playing basketball –
but when he retired from his job as an insurance executive and business
coach, he felt a pull toward a comfortable chair in front of the TV. But
that got boring fast, so when he saw a Help Wanted sign at Planet
Fitness, he applied and worked his way up to become a trainer.

[to top of second column]
|
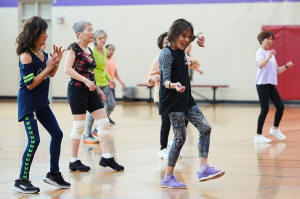
Instructor Bengie Santos, 72, leads a class for older adults,
including Pilar Madayag, left, at the Dale Turner Family YMCA, April
30, 2026, in Shoreline, Wash. (AP Photo/Lindsey Wasson)
 Most of his clients are 50 or older.
“I’ve heard people say I set a good example and I try to,” he says.
“At my age, if I can exercise, why can’t other people too?”
Recruiting older instructors
The YMCA in Seattle is mindful of the value of having an older
instructor leading a senior class.
“We don’t put an age limit on our applicants,” says program
executive Kerry Ashby, who started there as a younger instructor for
the Active Older Adults program. The Y does recruit from within the
older adult classes, and a lot of instructors are former
participants.
“We know it’s more than just fitness for the majority of our active
older adults,” Ashby says. “It’s also the mental and emotional
aspect.”
Tom Kleinecke, 67, has been a member of the Y for 18 years and has
sampled many different classes. Younger instructors, he says, tend
to teach their classes, while Santos leads and inspires.
Kleinecke says his fitness and endurance have improved noticeably
since he started taking three of Santos’ classes a week. Although
he’s fit, he says, it’s challenging to keep up with her.
One of the classes includes a faster-paced dance class not designed
for older students. At first, parts were tough to follow, he said,
but before long, he knew all the steps, from mambo to moonwalk.
“If you’re worried about being out of sync and uncoordinated, don’t
worry about it because nobody is paying attention to you,” Kleinecke
says. They’re all watching the instructor.
Now, he and his wife go out dancing on the weekends.
Looking forward to working out
Santos says she considers the ages and backgrounds of her students
when choosing the exercise class soundtrack.

“I always tell other instructors it isn’t your class, it’s their
class,” she says.
She also teaches smaller classes in three senior living facilities.
Those students range from 80 to 102.
“The caregivers wheel them out. I play their music. It triggers
their memory,” from Judy Garland to Louie Armstrong, Santos says.
“When they see me they think they’re going to party.”
Some even waltz in their chairs.
All contents © copyright 2026 Associated Press. All rights reserved |